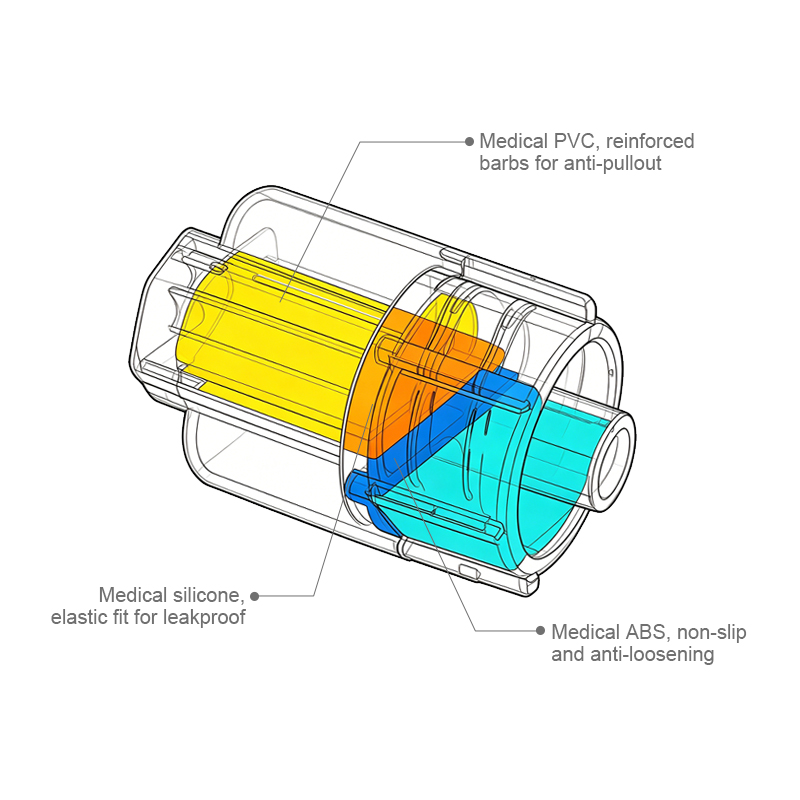
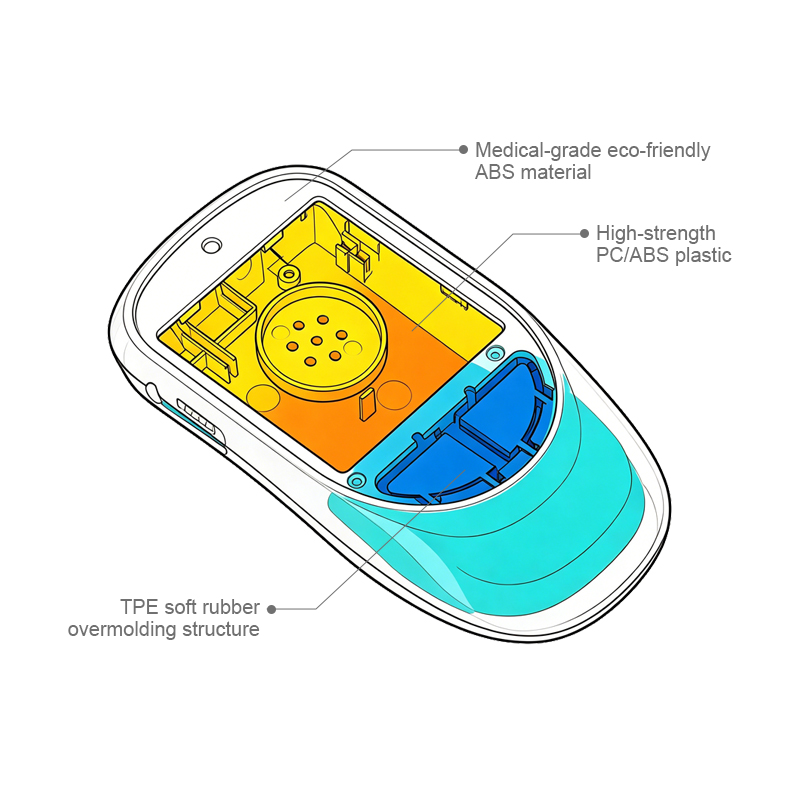
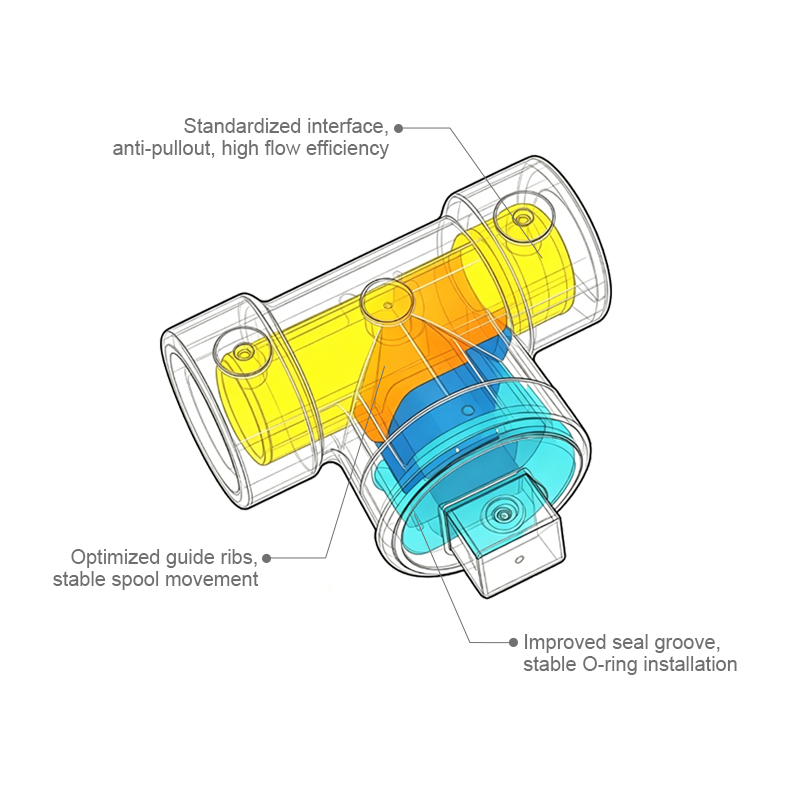
Nullam dignissim, ante scelerisque the is euismod fermentum odio sem semper the is erat, a feugiat leo urna eget eros. Duis Aenean a imperdiet risus.
Nullam dignissim, ante scelerisque the is euismod fermentum odio sem semper the is erat, a feugiat leo urna eget eros. Duis Aenean a imperdiet risus.


For a medical consumables client, the chemiluminescence reaction cups supplied by the original manufacturer suffered from long-term issues including substandard surface polishing, insufficient cleanliness and unstable precision dimensions. Defective products led to a low yield rate, impaired the accuracy of downstream experimental detection, hindered mass production progress, and failed to meet industrial compliance standards. After multi-party comparison and industry recommendation, the client selected our company for cooperation. Supported by years of experience in precision mold development and dust-free injection molding, we conducted a comprehensive analysis on existing defects. Optimizations were made in mold structure, surface treatment and production control to resolve quality problems. All inherent quality defects were fully eliminated. Stable product performance, improved yield and standardized mass production were achieved, steadily meeting the high cleanliness and high-precision requirements of medical chemiluminescence applications.
The client is a fast-growing medical consumables enterprise trapped in a supply chain dilemma. Large upstream manufacturers have solid capacity but rigid operations and tight production schedules. They respond slowly to customized adjustments, urgent orders and small-batch iterations, restricting the client’s development. Small workshops feature flexible communication and delivery, yet lack standardized management, complete quality control, traceability and medical compliance systems. They cannot meet strict dust-free production and audit standards, posing major compliance risks. Referred by industry peers, the client chose our cooperation. We fully meet the core needs of growing medical enterprises. With ISO management, dust-free workshops and full quality traceability, we satisfy high medical production requirements. Meanwhile, we offer flexible scheduling, stable lead time and high collaboration to support R&D, order changes and urgent mass production. We provide reliable custom mold and injection molding production with strict quality control. Complete audit materials, traceability records and compliance documents are supplied to help the client pass audits and secure compliance. Through long-term partnership, the client has expanded rapidly and built a new industrial base. We are honored to grow together and become their reliable core strategic partner.

The client, deeply entrenched in the industrial products sector, is urgently seeking a high-quality supplier capable of providing both custom mold fabrication and mass injection molding production. Its structurally complex industrial products required high-precision molding, yet the client’s former suppliers lacked advanced technology and sufficient capacity. Unable to resolve complex molding challenges and meet mass delivery demands, the client faced stalled R&D progress and missed market opportunities due to unstable processes and constant delivery delays. Amid tight project timelines, the client chose us after rigorous industry evaluations. Leveraging our profound precision mold expertise and one-stop supply chain system, we optimized tailored processes for complex product structures and streamlined the full workflow from mold design and sample testing to mass production. With strict full-process quality and production control, we effectively shortened lead times and guaranteed consistent product quality. We delivered premium industrial plastic components on schedule, fully resolving the client’s core issues: complex structure molding difficulties, mold-production disconnection and unmet urgent delivery needs. Our integrated customized production accelerated the client’s new product launch and helped them capture key market opportunities swiftly.
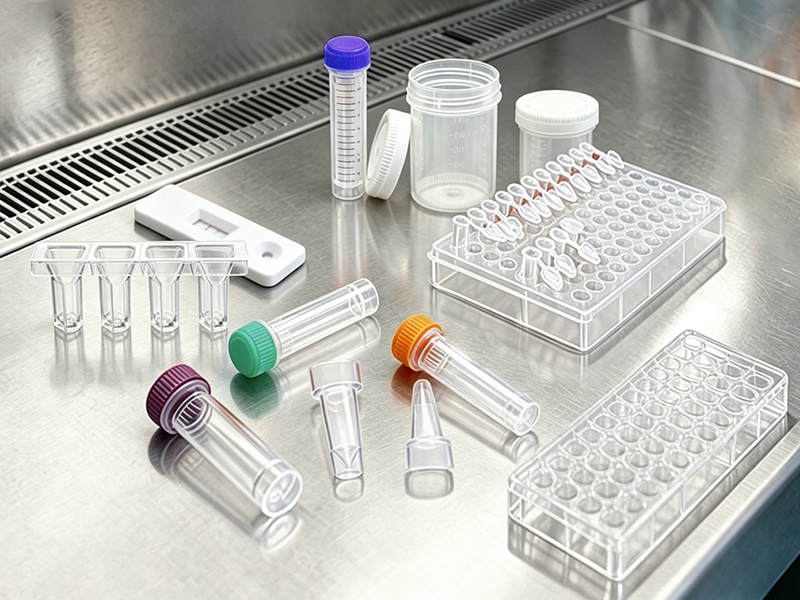
Medical-grade precision and clean production.

High-purity material for accurate bio-detection.

Ultra-precise dimensions for medical assembly needs.

Sturdy structure, weather-resistant and durable.

Micron tolerance for smart device precision assembly.

Precision injection molding, high structural strength.

Rayboen is a professional supplier with over 25 years of industry experience, specializing in the customized R&D and production of medical IVD consumables, as well as plastic housings and structural components for the medical, electronics and industrial sectors.Centering on precision mold design and manufacturing, plastic injection molding, and product structural analysis and optimization, Rayboen boasts extensive experience in material selection and diverse surface treatment capabilities, while providing one-stop supporting services including assembly, packaging, quality inspection and warehousing.With a professional team dedicated to R&D design, engineering, and manufacturing, Rayboen is committed to delivering reliable and highly cost-effective custom precision manufacturing solutions to customers in the IVD, medical, electronics, and industrial industries.
EXPERIENCE
EMPLOYEE
EQUIPMENT
FACTORY AREA